RESEARCH ARTICLE | https://doi.org/10.5005/jp-journals-10005-2037 |
Efficacy of Silver Diamine Fluoride on Streptococcus mutans Count Present in Saliva
1–4Department of Pediatric and Preventive Dentistry, Santosh Dental College and Hospitals, Ghaziabad, Uttar Pradesh, India
Corresponding Author: Sarbojit Chakraborty, Department of Pediatric and Preventive Dentistry, Santosh Dental College and Hospitals, Ghaziabad, Uttar Pradesh, India, Phone: +91 8342978658, e-mail: sarbojit.chakraborty5@gmail.com
How to cite this article Chakraborty S, Gupta N, Gambhir N, et al. Efficacy of Silver Diamine Fluoride on Streptococcus mutans Count Present in Saliva. Int J Clin Pediatr Dent 2021;14(5):700–704.
Source of support: Nil
Conflict of interest: Can be used as a preventive treatment modality
ABSTRACT
Aim and objective: Evaluate the efficacy and longevity of silver diamine fluoride (SDF) to inhibit the dental caries initiator bacteria; Streptococcus mutans count in saliva.
Study design: Twenty children having class I and II dental caries with no prior restorations were included in the study. Unstipulated saliva samples were collected before and immediately after application of SDF, 1, 3, and 6 months after application, and intergroup comparison was done.
Materials and methods: Silver diamine fluoride was applied on the cavitated surfaces with microbrush tip under isolation, then wait for 4 minutes and washed away all surfaces with water spray. Saliva was collected in a sterile saliva collection tube and sent for microbiological culture assessment. CFU/mL of S. mutans count was done with the Digital colony counter. For statistical analysis, paired T-test was performed.
Results: Silver diamine fluoride showed a significant drop in CFU/mL of S. mutans count at 1 month then 3 and 6 months accordingly.
Conclusion: Silver diamine fluoride can be used to prevent and arrest dental caries and the best result can be achieved by quarterly application.
Keywords: 38% SDF, Carious primary tooth, Silver diamine fluoride, Silver diamine fluoride, Streptococcus mutans.
INTRODUCTION
Dental caries is a local infection modified by carbohydrates that destroy a tooth’s hard tissues. It is a complicated illness characterized by the interplay of several variables, including host, agent, substrate, and time. This concept was explained and postulated by WD Miller’s chemo parasitic theory of dental caries formation. The theory suggested that the demineralization of the tooth surface takes place due to the acid produced by the bacteria present on the tooth surface.1
Streptococcus mutans, according to Van Houte,2 plays a substantial part in the formation of dental caries, although Loesche3 said that it is the main pathogen. Dental caries is now viewed as a biological interaction in which social, behavioral, psychological, and biological variables interact in a very complicated dynamic manner.3 If caries is to occur and progress, the aforementioned criteria must be met.
Mutans streptococci have all of these virulent characteristics, implying that they play a role in the caries progression. Streptococcus mutans responds to low pH environments by enhancing their rate of acid production and reducing the pH further lower, leading to a cariogenic plaque.4
Longitudinal investigations have revealed a relative increase in S. mutans in plaque specimens from carious tooth surfaces. Streptococcus mutans levels in saliva of children from various backgrounds were shown to correspond with patients’ caries activity levels.5
Caries activity is a multifactor diagnosis based on recent experience, lesion development, and the clinical presentation of the lesion or cavities. Data from clinical examinations and assessments of variables related to the disease are used to measure caries activity. These data on dental caries can be gathered by traditional visual inspection and probing, as well as through various objective detection methods that depend on mineral changes as a basis for evaluating caries activity and risk evaluation.
Since 1960, silver diamine fluoride (SDF) has been utilized in caries prevention as an effective cariostatic agent.6 Silver diamine fluoride is an alternative therapy for the prevention and treatment of dental caries. Silver diamine fluoride has been shown to raise the pH of biofilm, reduces the dentin demineralization, and has anti-microbial action against S. mutans. A recent study of SDF found that it is a reliable, effective, efficient, and “equitable” caries control agent that may be utilized to achieve the World Health Organization’s Millennium Goals and fulfill the standards for 21st-century medical care set by the US Institute of Medicine. The study has also shown that topical application of SDF in the management of caries is a simple, cost-effective, and non-invasive process.7
AIM AND OBJECTIVE
The purpose of this study was to assess the efficacy and performance of SDF in inhibiting the bacteria which cause dental caries; S. mutans count in saliva immediately after application of SDF, after 1, 3, and 6 months of each patient.
MATERIALS AND METHODS
The sample size was determined for this study to be 20. A total of 20 children aged 3–10 years who visited the Department of Peadiatric and Preventive Dentistry at Santosh Dental College and Hospital in Ghaziabad were chosen randomly based on the selection criteria.
Inclusion Criteria
- Patient age ranging from 3 to 10 years.
- Deciduous carious molars (classes I and II).
- Minimum of two carious teeth present to receive the ART technique.
- Healthy children without any systemic disease were included in the study.
Exclusion Criteria
- Early childhood caries, rampant caries patients.
- Class II, III, IV, V, VI type caries.
- Uncooperative patients.
- Patients requiring multiple dental treatments.
Consent was obtained from parents/guardians after informing them about the study and all queries were met. Materials and armamentariums used were: 38% SDF (Fagmin, Argentina), Mitis Salivarius Agar Media, Mitis Salivarius agar plate/loop, saliva collection tube microbrush tips, and cotton rolls.
Collection of Saliva Sample
- Patients were requested to remain quiet with their heads down and mouths open, allowing saliva to flow passively from the lower lip into graded sterile saliva collecting tubes, yielding roughly 1 mL of saliva in the first 30 seconds.
- As a control, baseline saliva samples were collected from all of the chosen individuals before the clinical procedure.
- One milliliter of collected saliva was placed in a tube containing 1 mL of thioglycollate transport medium. The tubes were sterilized by alcohol from outside and were put into an icebox and transported to the laboratory for sampling.
Application
Atraumatic restorative technique was performed on the dental caries class I and II on deciduous molars with the help of spoon excavators under cotton roll isolation. Gently carious part of the enamel was removed with spoon excavators. Vaseline was applied to the lips. 38% SDF was applied using a single tufted microbrush on all the surfaces of the cavity and wait for 4 minutes. After that, cavity surfaces were washed with water thoroughly and patients were asked to pool the saliva in the mouth and collect it in the sterile tube using a drooling technique (Fig. 1).
- Saliva samples were collected immediately after the intervention.
- Immediate postoperative saliva sample was collected for microbiological inoculation and culture was obtained on the same day.
Transport of Collected Samples
- The samples were transported to the laboratory in a sterile icebox to help maintain the viability of the test organism, immediately after collection and processed on the same day. The samples were stored at the temperature of −10°C to prevent the loss of viability of the test organism.
Inoculation and Incubation
The saliva sample was buffered in 0.05 M phosphate solution to a dilution of 1/20 and agitated on a vortex for 30 seconds. One hundred microliters of this solution were used for inoculation on Mitis Salivarius agar plates. The plates were then incubated at 37°C for 48 hours. After 48 hours, colony characteristics were studied and the numbers of colony-forming units of Mitis Salivarius (CFU/mL) of saliva were counted (Fig. 2).

Figs 1A to F: (A) Class 1 carious molar; (B) Pretreatment saliva collection; (C) ART caries removal; (D) After ART treatment; (E) After SDF application; (F) Immediate after treatment saliva collection

Figs 2A to E: (A) Baseline saliva sample in mitis salivarius agar plate for S. mutans CFU/mL count; (B) Immediate postoperative saliva sample in mitis salivarius agar plate for S. mutans CFU/mL count; (C) After 1-month saliva sample in mitis salivarius agar plate for S. mutans CFU/mL count; (D) After 3 months saliva sample in mitis salivarius agar plate for S. mutans CFU/mL count; (E) After 6 months saliva sample in mitis salivarius agar plate for S. mutans CFU/mL count
| Variable | Frequency (n = 20) (%) | |
|---|---|---|
| Age group | 3–4 years | 4 (20) |
| 4–5 years | 4 (20) | |
| 5–6 years | 3 (15) | |
| 6–7 years | 2 (10) | |
| 7–8 years | 2 (10) | |
| 8–9 years | 1 (5) | |
| 9–10 years | 4 (20) | |
Colony Counting
Colony counts were obtained with the use of a manual colony counter on a grid present on the colony counter. The count of each sample was obtained and tabulated.
Posttreatment Saliva Sample Collection
The sample collected after the completion of the treatment was also analyzed for the S. mutans count similar to the pre-treatment sample. Likewise, all the saliva samples—1, 3, and 6 months post-treatment samples were collected in sterile collection tubes and transported and inoculated on the same day of collection and the result was obtained after 48 hours.
RESULTS
Out of 20 children who have received SDF application according to their age group, 8 (40%) of them belong to 3–5 years of age group. Only 1 (5%) were from the age group of 8–9 years of age (Table 1 and Fig. 3). The mean and standard deviation of SDF on S. mutans count recorded at five different intervals (before application of the varnish/sealant, immediately after the application, 1 month after the application, and 3 months after the application, and 6 months after the application) from the samples of 20 children. The spread of the data, which is indicated by the standard deviation, is also significant in each group for all intervals (Table 2). The p value is calculated at a 95% confidence interval between the S. mutans counts of the samples taken before the application of SDF, immediately after the application of SDF, 1 month post-application, 3 months post-application, and 6 months post the application of SDF. There was a drop in S. mutans count on three times when samples were collected, and the magnitude of the decline was statistically significant (Table 3). Among the three occasions, the magnitude of the decrease in S. mutans count has a more pronounced effect in the first 3 months when compared with before the application and immediately after the application (Fig. 4).
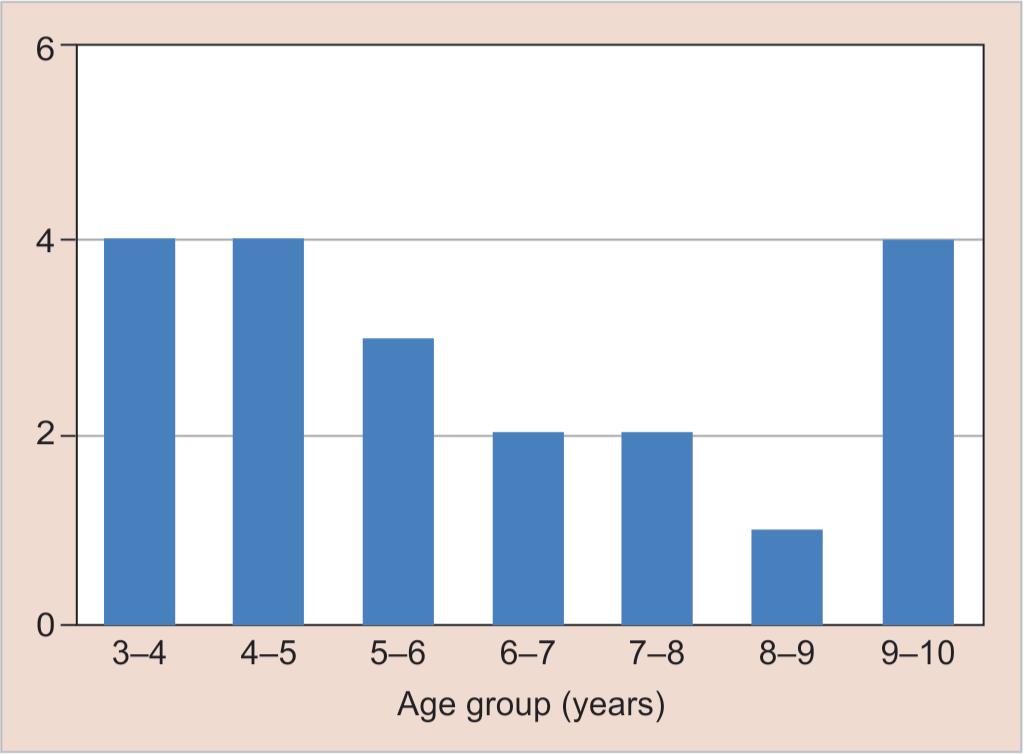
Fig. 3: Sample population according to age of SDF group
| Time of data collection | Silver diamine fluoride (n = 20) | |
|---|---|---|
| Before application | Mean ± SD | 406.80 ± (120.91) |
| Immediately after | Mean ± SD | 178.85 ± (66.65) |
| 1st month | Mean ± SD | 243.4 ± (71.89) |
| 3rd month | Mean ± SD | 292.35 ± (89.15) |
| 6th month | Mean ± SD | 341.75 ± (103.60) |
DISCUSSION
Dental caries is a truly complex disease, involving an interplay between a vulnerable host and a pathogen, i.e., the tooth, a cariogenic potential microflora, and a suitable local substrate to meet pathogenic flora requirements. Newbrun et al. applied a time factor dimension to the above three variables and named it the “Teratology of Caries.” Mutans streptococci are the most cariogenic pathogens because they are extremely acidogenic, generating short-chain carboxylic acids that destroy hard tissues like tooth enamel and dentin. S. mutans and S. sorbinus are the two most often isolated Mutans Streptococci species from saliva samples. It is more cariogenic to S. mutans than to S. sobrinus because S. mutans have unique cell-surface proteins.8 The primary attachment to the tooth is supported by S. mutans, while S. sobrinus lacks proteins of that kind. When both species coexist, the oral environment becomes more favorable to caries. Islam et al. stated that colonization of the tooth by S. mutans occurs immediately after the eruption.9 This eventually results in deep dental caries affecting the dentin and pulp if the colonization of fissures to their depths is pursued. Loesche et al. have stated that in the growth of caries, plaque, and biofilm, S. mutans have outnumbered many other species. Thus, to preserve oral health, successful eradication of the source of S. mutans is necessary. Research by HC Slavkin found a link between early childhood caries and S. mutans (1999), the most conspicuous bacterial culprit for their incidence.10
| Variables | Mean ± SD | p value (<0.05) |
|---|---|---|
| SDF (baseline) | 406.80 ± 120.9178 | 0.00005447991 |
| SDF (immediately after application) | 178.85 ± 66.6596 | |
| SDF (baseline) | 406.80 ± 120.9178 | 0.00000000131 |
| SDF (1 month) | 243.4 ± 71.89934 | |
| SDF (baseline) | 406.80 ± 120.9178 | 0.00000000189 |
| SDF (3 months) | 292.35 ± 89.15231 | |
| SDF (baseline) | 406.80 ± 120.9178 | 0.00000000324 |
| SDF (6 months) | 341.75 ± 103.6075 |
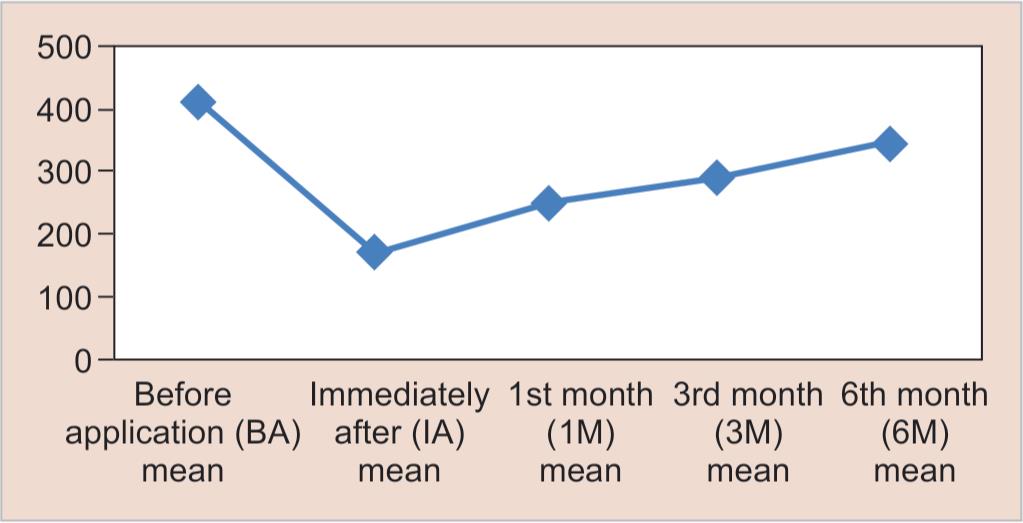
Fig. 4: Means of the Streptococcus mutans colony count after SDF application at various time intervals
Silver diamine fluoride a fluoride agent believed that it was capable of stopping the decay rate and at the same time preventing new caries from developing.11 For decades, SDF has been used worldwide to attempt and arrest primary and permanent dental caries. Since 1969, usage has been documented in Japan, Australia, Argentina, and other nations.12,13 A series of in vitro experiments were performed in Australia and Braga et al. proved its usefulness as caries and antibacterial agent in the United States.14 In 2014, SDF was cleared up in the US as a topical agent for the reduction in sensitivities by the US (FDA).15 In Nepal, Yee et al. successfully used SDF for caries arrest.12 It was suggested that the first therapeutic use of silver was around 1000 BC. The most recent use in medicine were silver nitrate, silver foil, and silver sutures, Naegeli16 considered silver nitrate to be an antimicrobial agent which was highly effective.12 Howe17 used silver nitrate for caries arrest for the next 50 years, with comparable findings.18 Craig et al. documented the dental use of the AgF solution in the 1970s.19 The Central Pharmaceutical Council of the Ministry of Health and Welfare of Japan has since the 1960s approved a related compound called SDF, as a rehabilitative agent in dentistry. Also in China, a 38% SDF solution was employed to avoid caries. In Southern California, few dentists used SDF to arrest early childhood caries. Projects for sub-Saharan Africa, Cuba, and other African countries were organized in group projects using the SDF to arrest caries.20 Silver diamine fluoride is a colorless aqueous solution with a pH of 10, 24.4–28.8% (253, 870 ppm) volume silver, 5.0–5.9% fluoride (44, 800 ppm), and ammonia containing silver ions and fluoride ions. Silver diamine fluoride is an allegedly more stable chemical and can be stored in a constant concentration. Fung et al. indicated that fluoride ions in SDF work specifically on the structure of the tooth while the silver phosphate is active against cariogenic bacteria. Silver diamine fluoride reacts with enamel hydroxyapatite and produces [Ca10(PO4)6F2] fluorapatite which is more acidic than hydroxyapatite resistant. The SDF has also shown that S. mutans have an antimicrobial effect.21
CONCLUSION
The findings of our study corroborate the folkloric prediction of SDF’s antibacterial efficacy in reducing the dental caries initiator S. mutans numbers in saliva. The best inhibitory effect was observed in 3 months, suggesting the quarterly application of SDF is an optimal choice.
The study result confirmed that SDF would be the most effective agent as not only bactericidal but also bacteriostatic actions that can control and eliminate the growth of S. mutans count in saliva.
CLINICAL SIGNIFICANCE
The current study suggests that in cases of incipient lesions, class I and class II cases the choice of treatment could be SDF application as a minimally invasive dentistry technique. Silver diamine fluoride can show bactericidal as well as bacteriostatic actions against dental caries causing bacteria S. mutans.
REFERENCES
1. Bowen WH, Birkhed D. Dental caries: dietary and microbiology factors. In: ed. L, Granath WD, McHugh ed. Systemized prevention of oral disease: theory and practice. Boca Raton (FL): CRC Press, Inc.; 1986. pp. 19–41.
2. Van Houte J. Role of microorganisms in caries etiology. J Dent Res 1994;73(3):672–681. DOI: 10.1177/00220345940730031301.
3. Loesche WJ. Role of Streptococcus mutans in human dental decay. Microbiol Rev 1986;50(4):353–380. DOI: 10.1128/mr.50.4.353-380.1986.
4. Marsh PD. The significance of maintaining the stability of the natural micro flora of the mouth. Br Dent J 1991;17(6):174–177. DOI: 10.1038/sj.bdj.4807647.
5. Chosak A, Cleaton-Jones P, Woods A, et al. Caries prevalence and severity in the primary dentition and Streptococcus mutans level in saliva of preschool children in South Africa. Community Dent Oral Epidemiol 1988;16(5):289–291. DOI: 10.1111/j.1600-0528.1988.tb01777.x.
6. Mattos-Silveira J, Floriano I, Ferreira FR, et al. New proposal of silver diamine fluoride use in arresting approximal caries: study protocol for a randomized controlled trial. Trials 2014;15(1):448. DOI: 10.1186/1745-6215-15-448.
7. Mei ML, Chu CH, Low KH, et al. Caries arresting effect of silver diamine fluoride on dentine carious lesion with S. mutans and L. acidophilus dual-species cariogenic biofilm. Med Oral Patol Oral Cir Bucal 2013;18(6):e824–e831. DOI: 10.4317/medoral.18831.
8. Newbrun E. Sugar and dental caries: a review of human studies. Science 1982;217(4558):418–423. DOI: 10.1126/science.7046052.
9. Islam B, Khan SN, Khan AU. Dental caries: from infection to prevention. Med Sci Monit 2007;13(11):RA196–RA203.
10. Slavkin HC. Streptococcus mutans, early childhood caries and new opportunities. J Am Dent Assoc 1999;130(12):1787–1792. DOI: 10.14219/jada.archive.1999.0138.
11. Rosenblatt A, Stamford TCM, Niederman R. Silver diamine fluoride: a caries “silver fluoride bullet”. J Dent Res 2009;88(2):116–125. DOI: 10.1177/0022034508329406.
12. Yee R, Holmgren C, Mulder J, et al. Efficacy of silver diamine fluoride for arresting caries treatment. J Dent Res 2009;88(7):644–647. DOI: 10.1177/0022034509338671.
13. Featherstone JD, Horst JA. Long used in other countries, silver diamine fluoride is now cleared for use in the U.S. While there is an assumption of use for pediatric caries control, here we explore indications for adults. J Multidisciplin Care Decis Dentis 2015. 1–8.Retrieved from Pubmed.
14. Braga MM, Mendes FM, De Benedetto MS, et al. Effect of silver diammine fluoride on incipient caries lesions in erupting permanent first molars: a pilot study. J Dent Child 2009;76(1):28–33.
15. Nelson T, Scott JM, Crystal YO, et al. Silver diamine fluoride in pediatric dentistry training programs: survey of graduate program directors. Pediatr Dent 2016;38(3):212–217.
16. Naegeli C. On the oligodynamic phenomenon in living cells. Denkschriften der Schweizerischen Naturforschenden Gesellschaft 1893;33:174–182.
17. Howe PR. A method of sterilizing and at the same time impregnating with a metal affected dentinal tissue. Dent Cosm 1917;59:891–904.
18. Gao SS, Zhao IS, Duffin S, et al. Revitalising silver nitrate for caries management. Int J Environ Res Public Health 2018;15(1):80. DOI: 10.3390/ijerph15010080.
19. Craig G, Powell K, Cooper M. Caries progression in primary molars: 24-month results from a minimal treatment programme. Community Dentis Oral Epidemiol 1982;9(6):260–265. DOI: 10.1111/j.1600-0528.1981.tb00342.x.
20. Shah S, Bhaskar V, Venkatraghavan K, et al. Silver diamine fluoride: a review and current applications. J Adv Oral Res 2014;5(1):25–35. DOI: 10.1177/2229411220140106.
21. Fung MHT, Wong MCM, Lo ECM, et al. Arresting early childhood caries with silver diamine fluoride-a literature review. J Oral Hyg Health 2013;1(03):117. DOI: 10.4172/2332-0702.1000117.
________________________
© The Author(s). 2021 Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and non-commercial reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.